Developing CRAC Channel
Inhibitors for Serious Acute
and Chronic Inflammatory,
Immunologic, and Cardiopulmonary
Diseases with High Unmet Need
Auxora™’s Dual Mode of Action: Modulating the Immune Response and Helping Protect Against Tissue Cell Injury
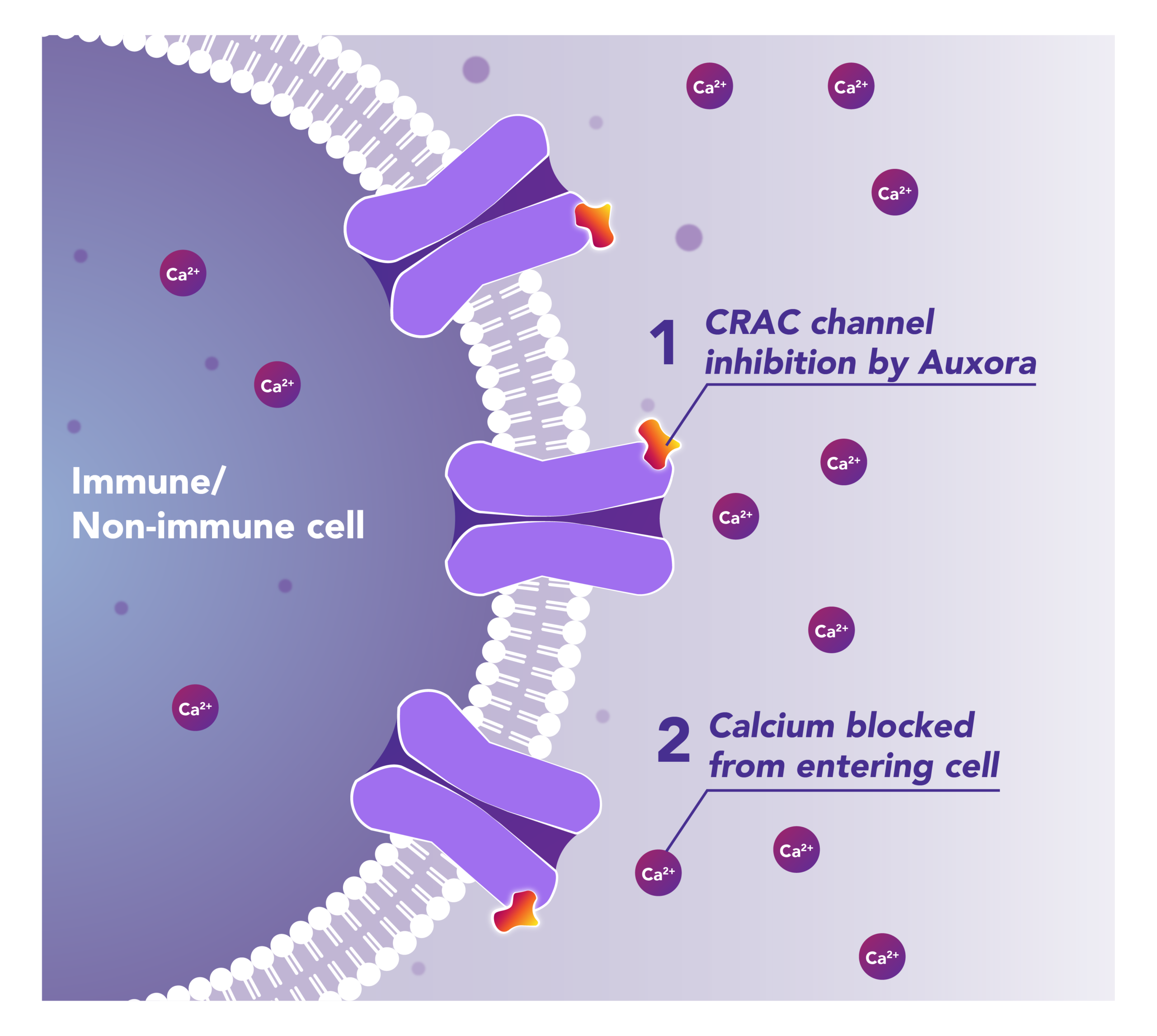
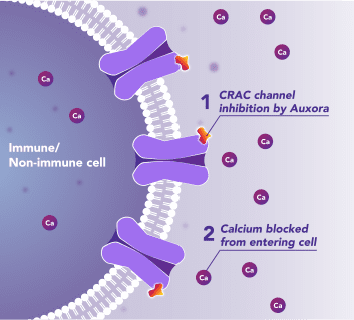
Preclinical and clinical data have demonstrated that the inhibition of disease-activated CRAC channels may have a therapeutic effect through a dual mechanism involving both anti-inflammatory activity and tissue cell protection.
CalciMedica is developing Auxora™ as a potential disease-modifying therapy for patients with acute, life-threatening inflammatory and immunologic diseases with high unmet need.
CalciMedica is also advancing CM5480, a next-generation CRAC channel inhibitor, for chronic inflammatory, immunologic, and cardiopulmonary diseases.
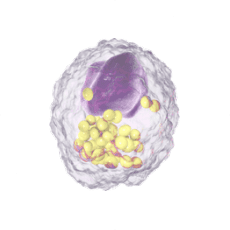
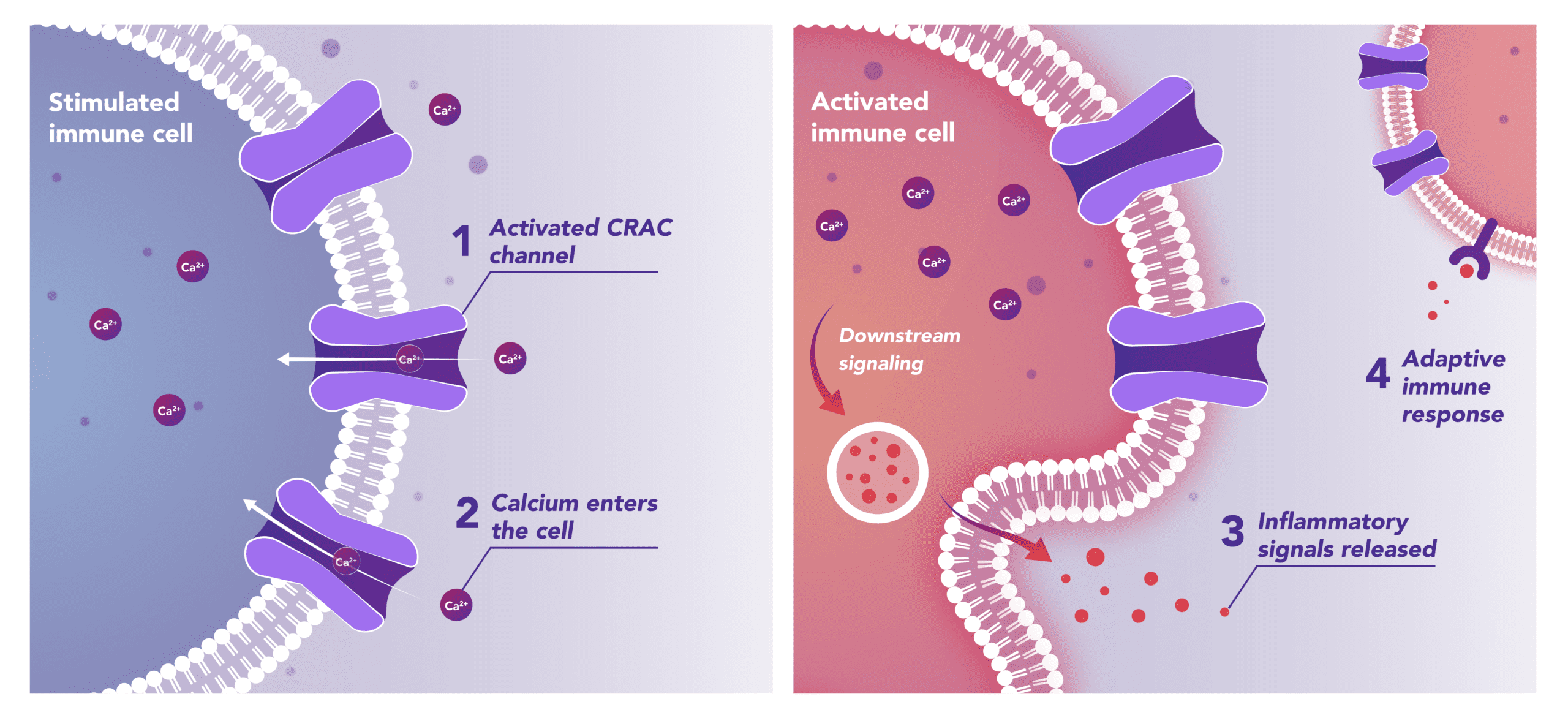
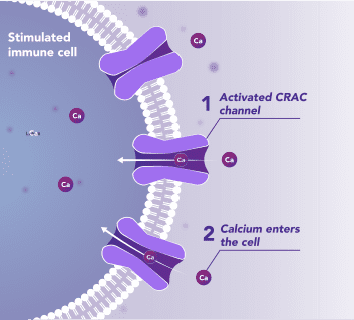
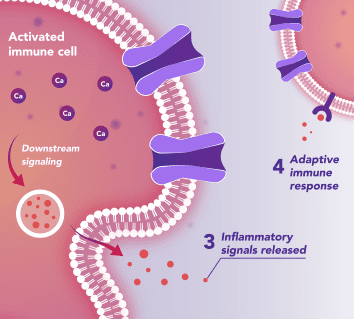
Immune Cells:
Modulates Immune Response
In immune cells, activation of CRAC channels in response to disease or trauma is a key step in initiating the adaptive immune response and the generation of inflammatory mediators such as cytokines. CRAC channels also contribute to activation of cells involved in the innate immune response. Inhibiting CRAC channels may reduce immune cell activation and the inflammation it drives.
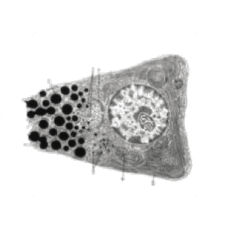
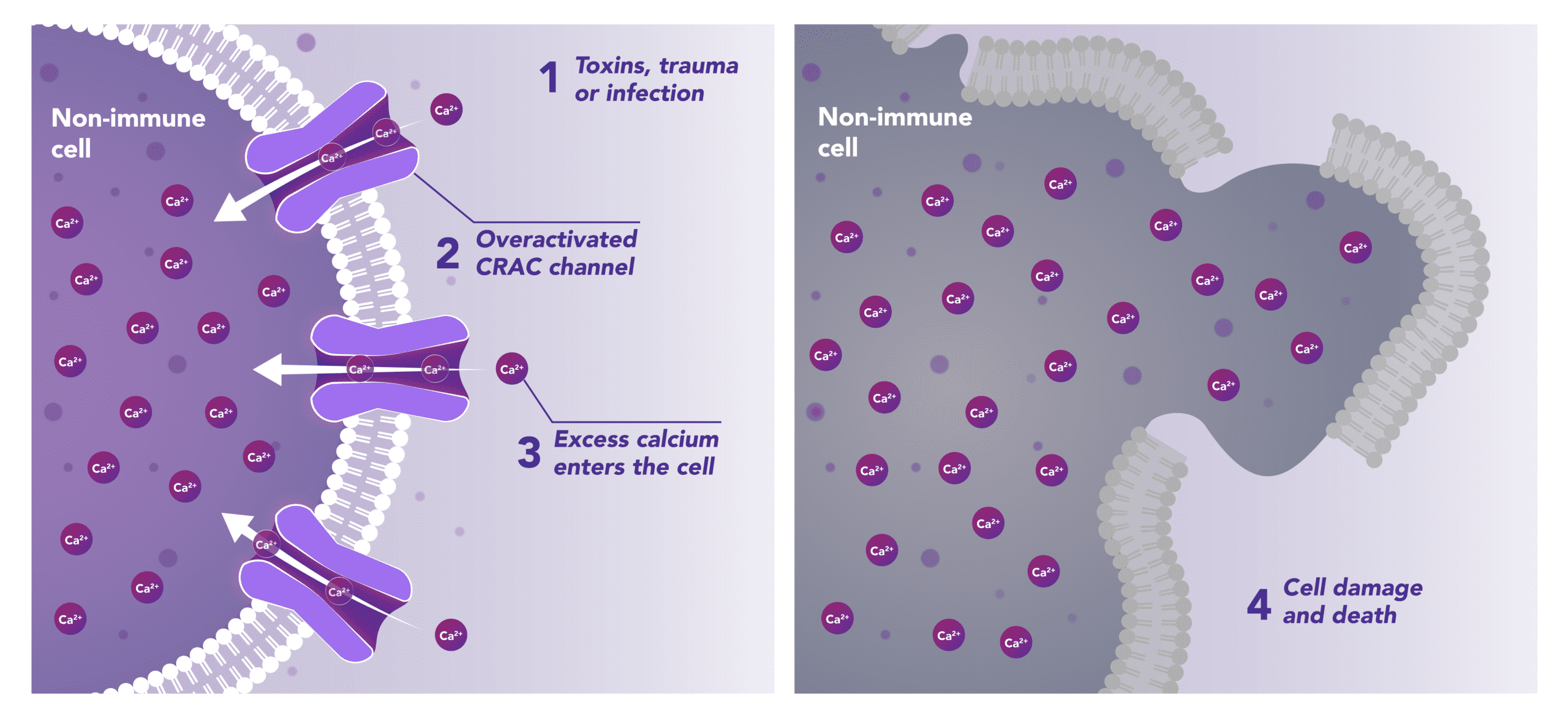
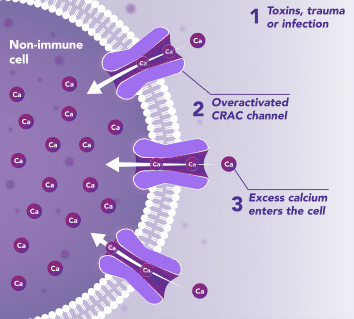
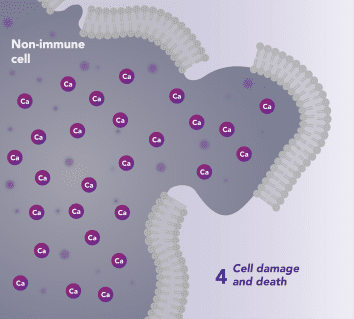
Non-Immune Cells:
Limits Excess Calcium Entry
In affected organ tissue cells, CRAC channels can become inappropriately activated or overactivated, resulting in excess calcium entry. This calcium overload can cause cellular injury and necrosis or trigger apoptosis pathways leading to programmed cell death. Inhibiting CRAC channels may help reduce organ damage by limiting excess calcium influx.
“Since the founding of CalciMedica, we have advanced research on CRAC channel inhibitors and their role in acute and chronic inflammatory, immunologic, and cardiopulmonary diseases. Our pipeline includes Auxora, our proprietary intravenous CRAC channel inhibitor, and CM5480, our next-generation CRAC channel inhibitor designed for chronic dosing. Together, these programs reflect our commitment to developing differentiated therapies for diseases with high unmet need.”

CalciMedica’s lead pipeline candidate, Auxora™, is a proprietary intravenous small molecule CRAC channel inhibitor. Across multiple studies in critical care settings, Auxora™ has shown clinical activity together with a favorable safety profile.
The Company’s next-generation CRAC channel inhibitor, CM5480, is in preclinical development for the treatment of chronic inflammatory, immunologic diseases, and cardiopulmonary diseases.
| Program1 | Indication | Phase of Development | Anticipated Milestones | |||
| Preclinical | Phase 1 | Phase 2 | Phase 3 | |||
| Acute Disease (IV) | ||||||
| Auxora | Acute Pancreatitis | Final pivotal trial design 1H 2026 | ||||
| Auxora | Acute Kidney Injury with Respiratory Failure |
FDA discussions: 2Q 2026 |
||||
| Auxora | Asparaginase-Induced Pancreatic Toxicity in Pediatric Patients | Update – 2026 | ||||
| Chronic Disease (Oral) | ||||||
| CM5480 | Pulmonary Arterial Hypertension | Add’l preclinical data – 2H 2026 Initiate Ph1 – 2027 |
||||
with Respiratory Failure
Initiate Ph1 – 2027
AP hospitalizations per year in the US, including approximately 170K with predicted severe disease
- Standard of care: Supportive care, including IV fluids, pain control, and nutrition
- Severe complications include pancreatic necrosis, life-threatening organ failure, and acute respiratory distress syndrome (ARDS)
- Major economic burden: >1M hospital days annually and >$3B in annual US cost
ICU patients per year in the US with Stage 2 or 3 AKI, including approximately 800K with concurrent respiratory failure
- Standard of Care: Supportive care, management of complications, and dialysis
- AKI can progress to chronic kidney disease, end-stage kidney disease, and death
- 90-Day Mortality: ~50%
Adults with PAH in the US
- Standard of care: currently approved therapies target disease-related pathways other than Orai1 and are not curative
- PAH causes chronic elevation in pulmonary artery pressure and pulmonary vascular resistance, which can lead to right ventricular (RV) hypertrophy, RV dysfunction, RV failure, and death
CalciMedica’s Proprietary CRAC Channel Inhibition Technology for Inflammatory, Immunologic, and Cardiopulmonary Diseases with High Unmet Need
“We have generated encouraging findings with our lead compound, Auxora™, including reductions in severe organ failure, improvements in recovery-related outcomes, shorter hospital stays, and fewer downstream complications in selected settings. These results support our continued mission to develop therapies for acute and chronic inflammatory, immunologic, and cardiopulmonary diseases where current treatment options remain insufficient.”