
Developing Novel Therapies
for Serious Acute and
Chronic Inflammatory,
Immunologic, and
Cardiopulmonary Diseases
CalciMedica is a clinical-stage biopharmaceutical company developing novel therapies based on CRAC (calcium release-activated calcium) channel inhibition therapies for serious acute and chronic inflammatory, immunologic, and cardiopulmonary diseases.
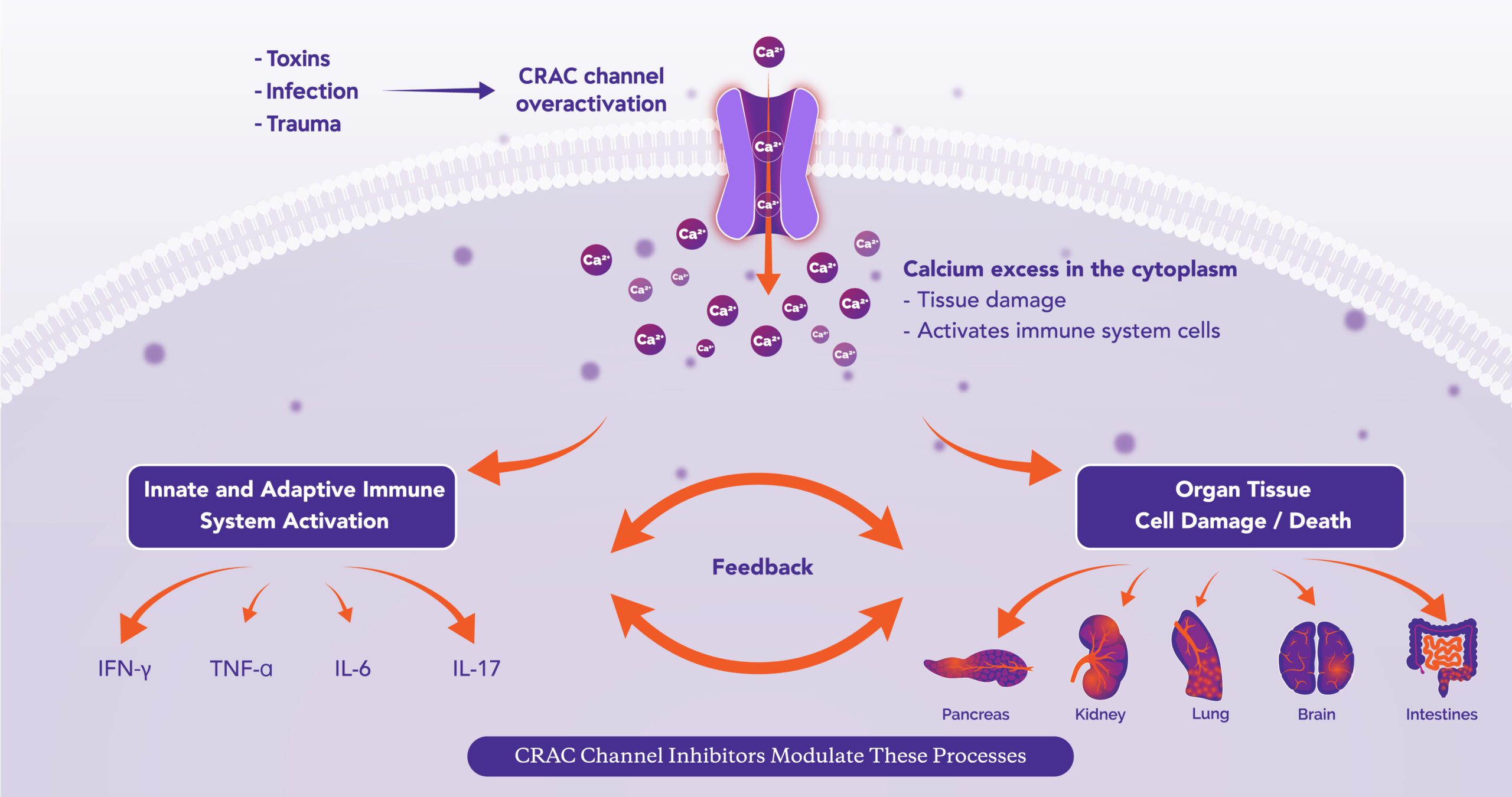
The Leader in CRAC Channel Inhibition: We are advancing a new class of therapies designed to modulate the immune response and help protect against tissue cell injury, two processes central to many serious acute and chronic diseases.
Our lead compound, Auxora™, has the potential to address
broad unmet need in critical care by improving outcomes
in life-threatening conditions for which there are
no-approved disease-modifying treatments.
AP hospitalizations per year in the US, including approximately 170K with predicted severe disease
- Standard of care: Supportive care, including IV fluids, pain control, and nutrition
- Severe complications include pancreatic necrosis, life-threatening organ failure, and acute respiratory distress syndrome (ARDS)
- Major economic burden: >1M hospital days annually and >$3B in annual US costs
ICU patients per year in the US with Stage 2 or 3 AKI, including approximately 800K with concurrent respiratory failure
- Standard of Care: Supportive care, management of complications, and dialysis
- AKI can progress to chronic kidney disease, end-stage kidney disease, and death
- 90-Day Mortality: ~50%
Our other proprietary drug candidate, CM5480, is a next-generation CRAC channel inhibitor designed for chronic dosing.
Our other proprietary drug candidate, CM5480, is a next-generation CRAC channel inhibitor designed for chronic dosing.
Adults with PAH in the US
- Standard of care: currently approved therapies target disease-related pathways other than Orai1 and are not curative
- PAH causes chronic elevation in pulmonary artery pressure and pulmonary vascular resistance, which can lead to right ventricular (RV) hypertrophy, RV dysfunction, RV failure, and death

Auxora™
Auxora™, an intravenous formulation of the proprietary
small molecule (CRAC) channel inhibitor zegocractin, is
being developed for the treatment of multiple
acute inflammatory and immunologic illnesses.


A Leadership Team
Dedicated to Science and
Innovation.
The CalciMedica team includes scientists, physicians,
drug development experts, and entrepreneurs dedicated to
advancing CRAC channel inhibitors for serious acute and chronic
inflammatory, immunologic, and cardiopulmonary diseases.


“Our focus at CalciMedica is on developing and bringing to market a new class of therapies – CRAC channel inhibitors – to treat serious inflammatory, immunologic, and cardiopulmonary diseases. We are committed to addressing areas of high unmet need with the goal of delivering safe and effective therapies to patients whose current treatment options remain inadequate.”

