
Acute Pancreatitis
Acute Pancreatitis represents a major unmet need in critical care medicine, with no approved disease-modifying therapies.
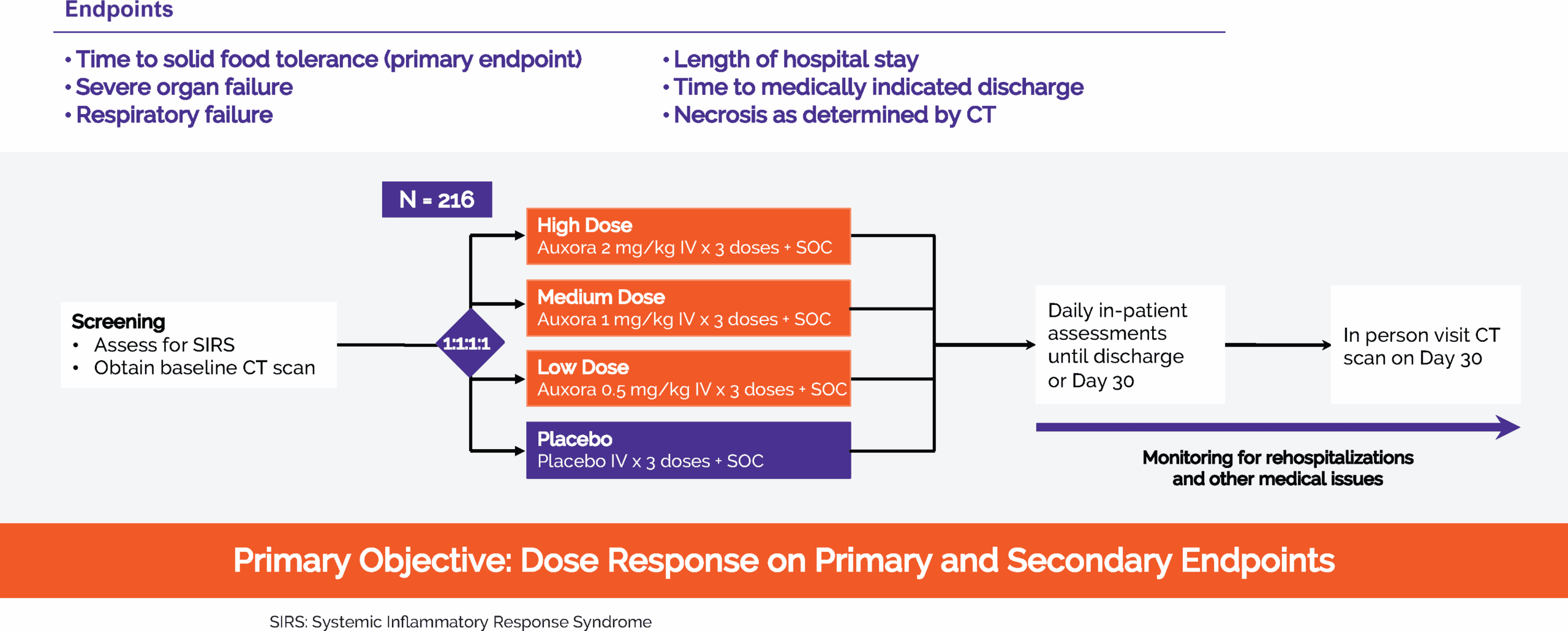
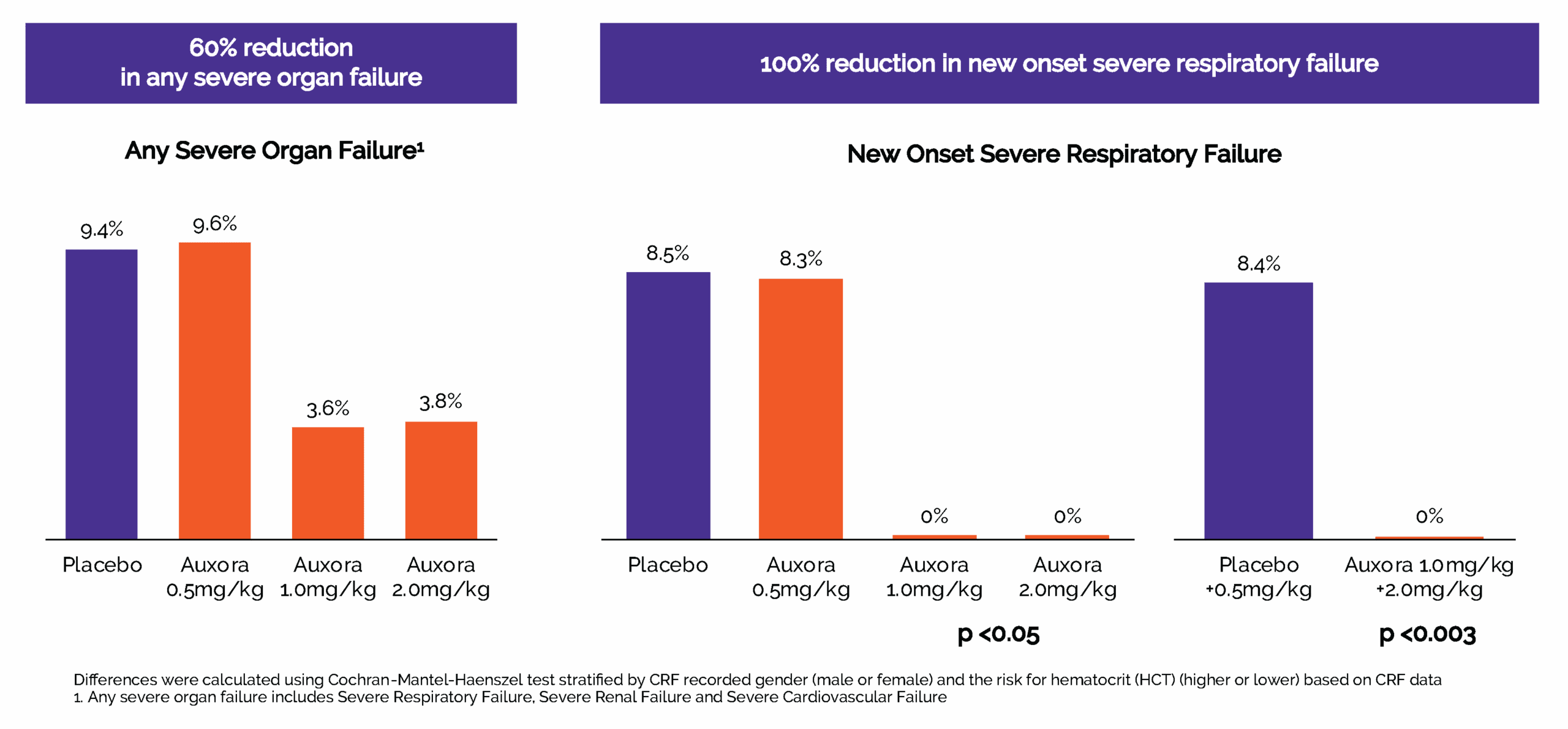
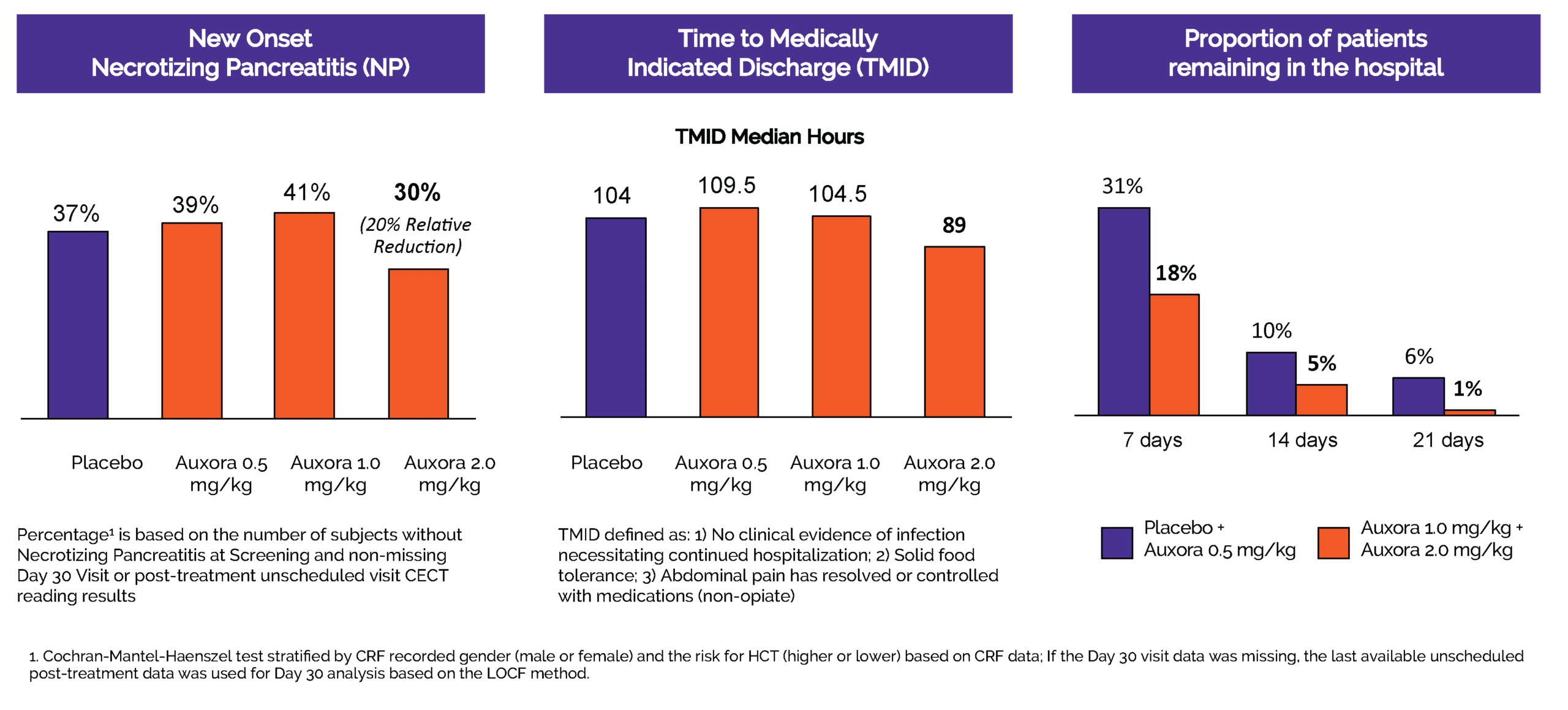
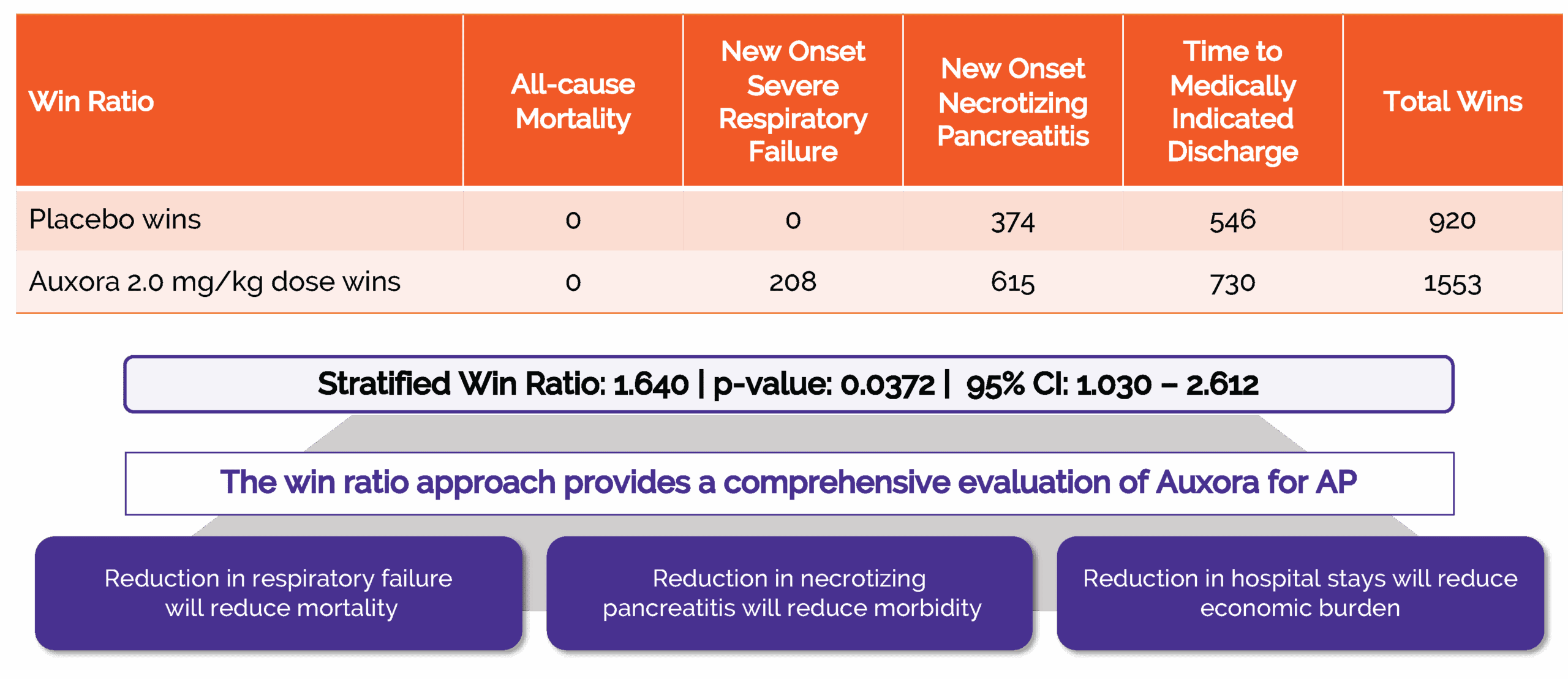
In 2024, we completed CARPO, an international, randomized, double-blind, placebo-controlled Phase 2b trial evaluating AuxoraTM versus placebo in 216 patients with acute pancreatitis (AP) and accompanying SIRS. The trial met its primary objective, demonstrating a statistically significant dose response in median time to solid food tolerance in a prespecified subgroup of patients with hyper-inflamed AP. In addition, both the medium- and high-dose Auxora arms showed reductions in severe organ failure and a 100% relative risk reduction versus placebo in new-onset severe respiratory failure. This effect was statistically significant for the high- and medium-dose arms individually (p<0.05) and for the combined high- and medium-dose Auxora patients compared with the combined placebo and low-dose Auxora patients (p=0.0027). The high-dose arm also showed an approximately 20% relative risk reduction in new-onset necrotizing pancreatitis versus placebo, a 15-hour shorter median time to medically indicated discharge, and a stratified win-ratio of 1.640 (p=0.0372) across selected key endpoints.
Reduction in new-onset respiratory failure
Reduction in severe organ failure
Reduction in necrotizing pancreatitis
Reduction in hospital stays > 21 days


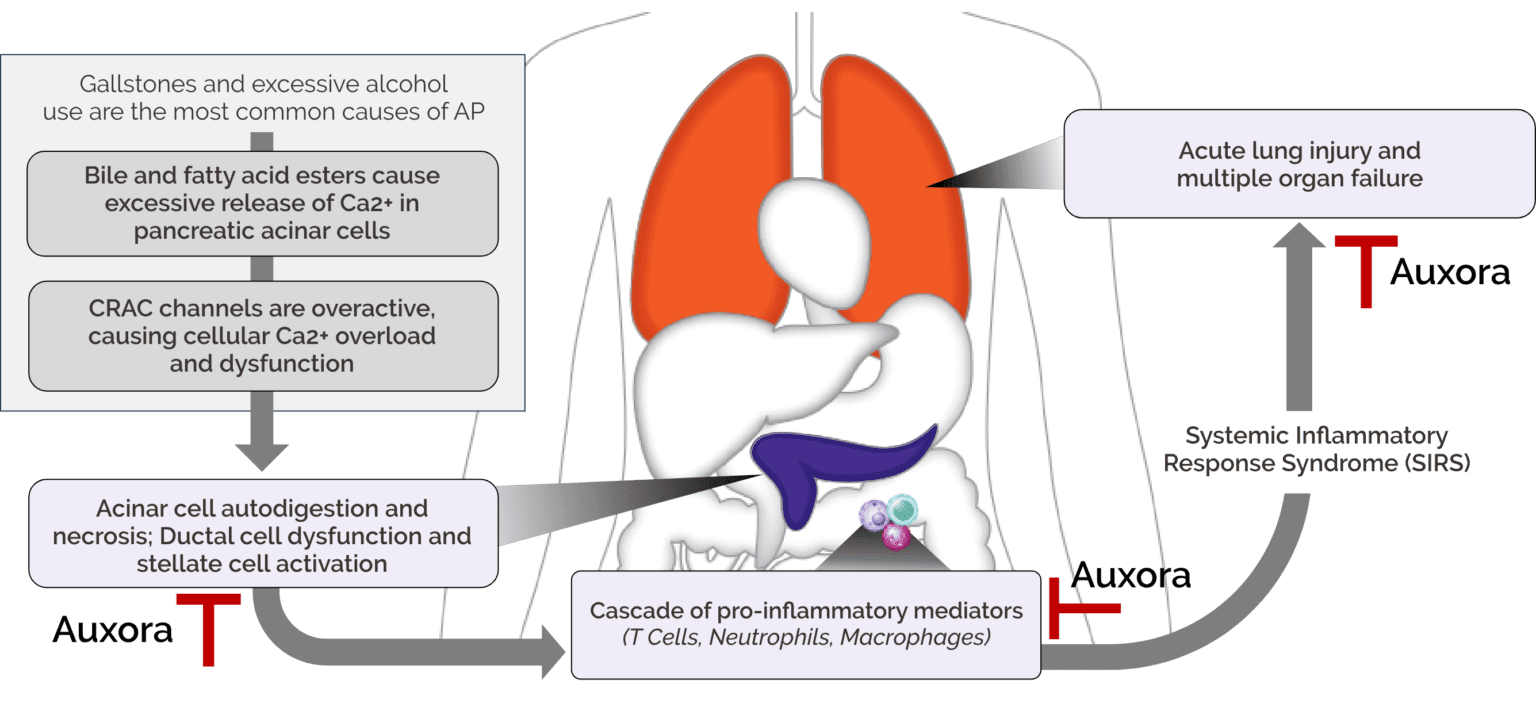
Acute pancreatitis (AP) is an acute inflammatory disease of the pancreas that typically presents with severe upper abdominal pain, often accompanied by nausea and vomiting. During an episode of AP, inflammation can lead to pancreatic cell death, tissue necrosis, and systemic inflammation. Normal pancreatic function, including secretion of digestive enzymes into the gut, can be disrupted.


A central challenge in AP is that severe systemic complications can arise from the inflammatory response triggered by pancreatic injury. Systemic inflammatory response syndrome (SIRS), driven by pancreatic tissue damage, can amplify inflammation and contribute to failure of organs beyond the pancreas, including the lungs, kidneys, and heart. Patients who present with risk factors such as SIRS and substantial pancreatic inflammation are more likely to progress to life-threatening severe AP.
There are currently no approved disease-modifying therapeutics for AP.
Hospitalizations per year for AP
~170K hospitalizations per year for AP with SIRS
~60K develop severe disease
>1M hospital days annually due to complications including organ failure, pancreatic necrosis, and mortality
Auxora in Acute Pancreatitis
Our Clinical Studies in AP
Acute Pancreatitis Phase 2a Clinical Trial
Study Design
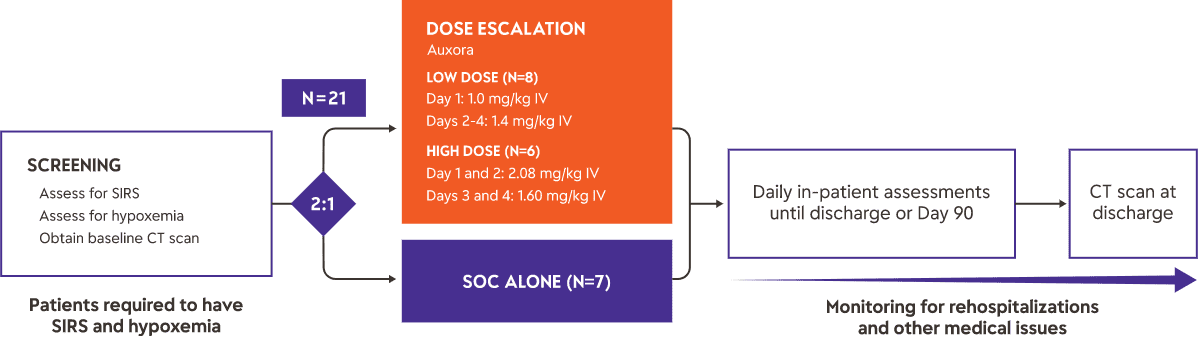
We completed a Phase 2a clinical trial of Auxora™ in 21 patients with AP and predicted severe disease based on the presence of SIRS and hypoxemia. Fourteen patients received Auxora™ plus standard of care—typically aggressive fluid resuscitation and pain management—at two dose levels, and seven received standard of care alone. Auxora™ was administered once daily by IV for up to four days. As an early proof-of-concept study, this trial was not powered for statistical significance on its endpoints.
Results
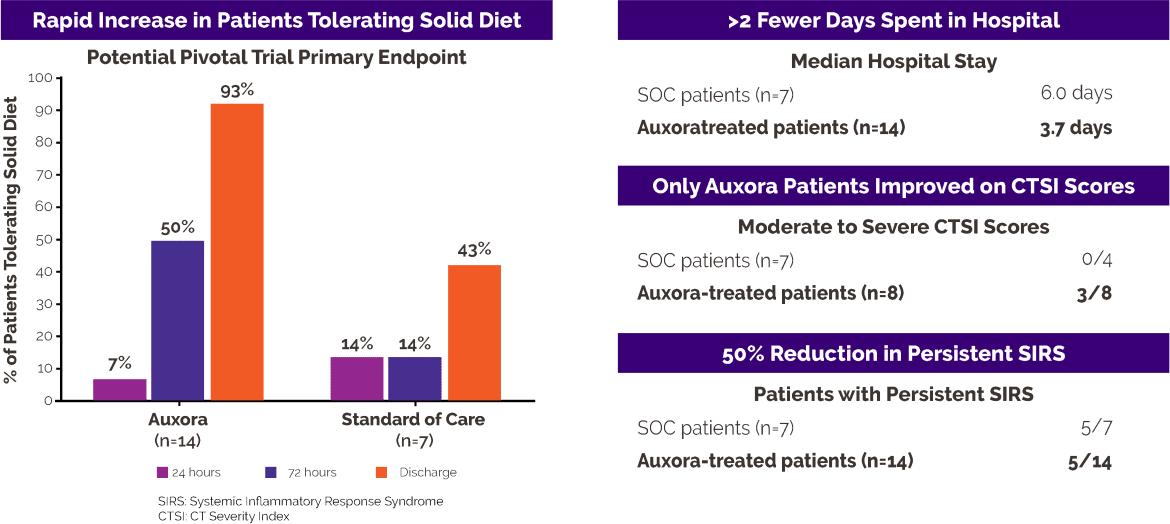
Severe upper abdominal pain is a hallmark symptom of AP, and patients are often unable to tolerate solid food until the pancreatitis begins to resolve. At study entry, one patient in the Auxora™ group (7%) and one patient in the standard-of-care group (14%) were tolerating solid food. After 72 hours, seven of 14 Auxora™ patients (50%) were tolerating solid food compared with one of seven patients receiving standard of care alone (14%). At hospital discharge, 13 of 14 Auxora™ patients (93%) could tolerate solid food compared with three of seven patients receiving standard of care alone (43%).
Patients treated with Auxora™ were discharged after a median hospital stay of 3.7 days, compared with 6.0 days for patients receiving standard of care alone.
Persistent SIRS, defined as SIRS lasting more than 48 hours, is an indicator of continued inflammatory activation and is predictive of organ failure in AP. All patients in this study had SIRS at enrollment. Persistent SIRS developed in five of 14 patients (36%) treated with Auxora™, compared with five of seven patients (71%) treated with standard of care alone.
For more details on the clinical trial, please visit: A Study of Auxora in Patients With Acute Pancreatitis and Accompanying SIRS.
Information on our Expanded Access Policy can be obtained through the following link.
CARPO Phase 2b clinical trial in patients with AP and accompanying SIRS
Study Design
The target population for Auxora™ in the completed Phase 2b CARPO clinical trial was AP patients with accompanying SIRS, which we estimate to be approximately 170,000 patients per year in the United States.
For more details on the clinical trial, please visit: A Study of Auxora in Patients with Acute Pancreatitis and Accompanying SIRS (CARPO).
Information on our Expanded Access Policy can be obtained through the following link.
CARPO Phase 2b clinical trial in AP with SIRS
Results
Auxora high and medium-doses reduced severe organ failure and completely prevented new-onset severe respiratory failure
Auxora high dose showed improvements in key secondary endpoints
Integration of key endpoints into Win Ratio demonstrated potential benefits of Auxora high dose compared to placebo


Oral CRAC channel inhibitors.
Solving The Unmet Need For
Patients With Chronic
Inflammatory Disease, such as
Chronic Pancreatitis
Beyond Auxora, we are also developing CRAC channel inhibitors for use in chronic inflammatory indications such as chronic pancreatitis that would be administered as oral agents. We are currently conducting IND-enabling preclinical testing on several CRAC channel inhibitors for use in chronic inflammatory diseases.
Beyond Auxora, we are also developing CRAC channel inhibitors for use in chronic inflammatory indications such as chronic pancreatitis that would be administered as oral agents. We are currently conducting IND-enabling preclinical testing on several CRAC channel inhibitors for use in chronic inflammatory diseases.

