
Auxora for the Treatment
of Acute Kidney Injury
with Respiratory Failure
The rationale for Auxora in AKI is supported by both human clinical and preclinical data. In our severe and critical COVID-19 pneumonia trial (CARDEA), we observed a nearly 40% reduction in reported AKI among Auxora-treated patients compared with placebo-treated patients who did not have kidney injury at enrollment. In a post hoc analysis of CARDEA patients with compromised kidney function at enrollment, Auxora-treated patients had a 63% relative reduction in mortality compared with placebo-treated patients. Consistent with other studies in AKI with respiratory failure, placebo patients had a mortality rate of nearly 50%. Biomarker analyses from CARDEA were also supportive of a potential treatment effect on the kidney and correlated with improved outcomes.
Preclinical studies supporting human clinical development showed that treatment of rats with AKI with Auxora improved estimated glomerular filtration rate (eGFR) by 61%, decreased infiltration of mononuclear inflammatory cells into the kidney by 30%, and reduced mortality versus placebo.
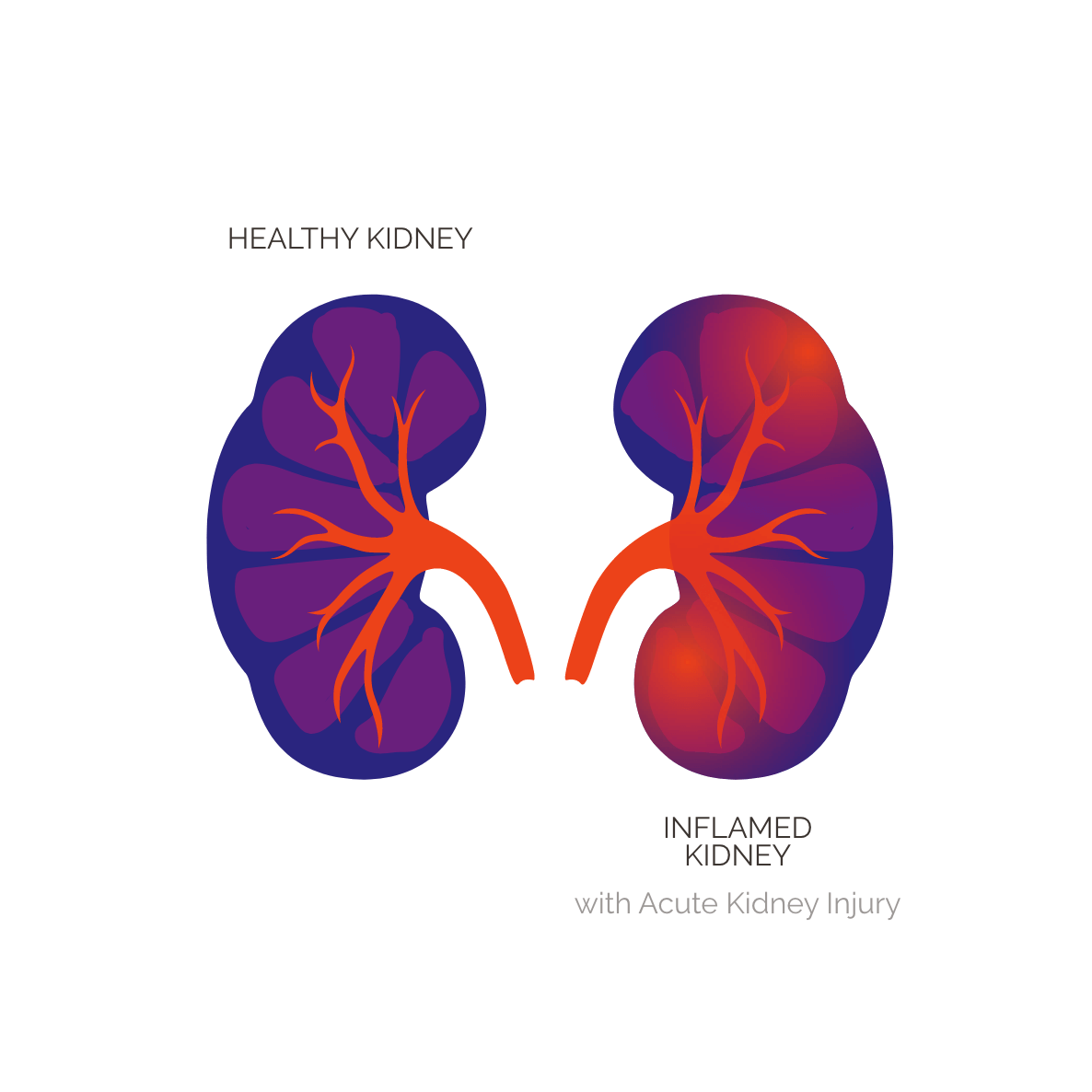

AKI is a sudden decline in kidney function, reflected by increased serum creatinine and/or reduced urine output. It can occur as a complication of other serious illnesses, including sepsis, respiratory infection and failure, acute pancreatitis, trauma, surgery, and burns. AKI is marked by three phases: the initial injury, progression or recovery from that injury, and the long-term damage that may follow.
AKI is classified as stage 1, 2 or 3 depending on severity. In the setting of respiratory failure, stage 2 and stage 3 AKI are associated with approximately 50% or greater risk of death during hospitalization and in the 90 days after discharge. Patients who experience AKI are also at risk for long-term complications, including chronic kidney disease and end-stage kidney disease. In the United States, approximately 1.25 million ICU patients each year have stage 2 or stage 3 AKI, and roughly 800K of those patients also have concurrent respiratory failure. There are currently no approved therapies for AKI.


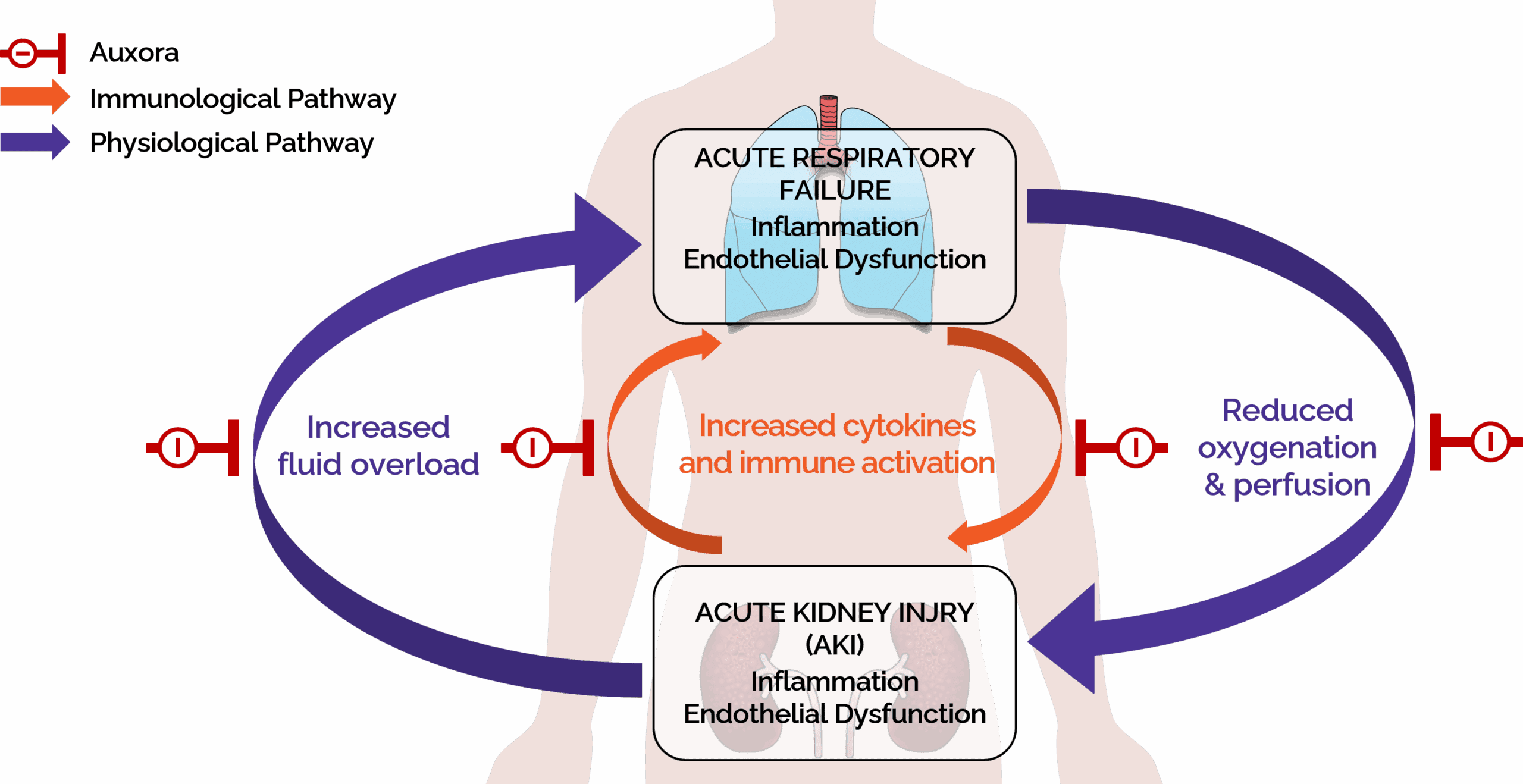
Auxora targets key pathways driving AKI with respiratory failure


patients per year in the US with AKI
of these patients have stage 2 or stage 3 (moderate to severe) AKI, of whom 800K also have respiratory failure
- Chronic kidney disease
- End-stage kidney disease
- Dialysis and/or kidney transplant
- Death
Auxora targets key pathways driving AKI with respiratory failure




The Phase 2 KOURAGE trial evaluating Auxora in patients with Stage 2 or Stage 3 AKI with associated respiratory failure (AHRF) has been discontinued following a recommendation from the trial’s Independent Data Monitoring Committee. The Committee did not identify evidence of drug-related toxicity, and the Company’s comprehensive review, performed in conjunction with external experts, reached the same conclusion. Imbalances in the patients’ severity of disease at baseline may have contributed to the observed safety concern. The Company plans to discuss the KOURAGE data and potential future development in AKI with the FDA.



